August 6, 2018
The rapid expansion of T-cell and other cell-based therapies has ignited discussions about how clinicians and researchers harvest, culture, process or directly alter therapeutic cells. The guest editors of a new SLAS Technology Special Issue delve into innovative tools that improve the process by scaling up manufacturing, reducing costs and improving safety.

“Immunotherapy is a hot bed of activity,” says Reginald Smith, Ph.D., a principal scientist at the General Electric Global Research Center, (GE; Niskayuna, NY, USA). “The biggest spotlight right now is on the 2017 Federal Drug Administration (FDA) approval of chimeric antigen receptor (CAR) T-cell therapies for Novartis International AG (Basel, Switzerland) and Kite Pharma (Santa Monica, CA, USA).” This action, according to a report released in May 2018 from the Cancer Research Institute (New York, NY), prompted incredible growth in the immunotherapy space with the entry of 350 new cancer cell therapies into the global development pipeline since September 2017, representing an 87 percent increase in less than nine months.
A potential stumbling block to this growth is the complexity of the manufacturing process, which includes selection of peripheral blood mononuclear cells from whole blood, activation of T-cells, transduction with CAR viral vectors or transposons and expansion in an appropriate bioreactor. “For robust manufacturing to work, the multiple steps and procedures performed on these cells must be integrated in a better fashion,” says Christopher Puleo, Ph.D., a biomedical engineer at GE.
Brian Davis, Ph.D., a GE cell biologist and a veteran of cell and gene therapy since 1994, says that cell processing needs a meeting of the minds. “On one hand you have the academics who are developing these therapies and on the other you have companies that are industrializing therapies. We need to merge the two,” he explains.
“Conventionally a lot of people start off doing small-scale protocols that are performed by graduate students or laboratory technicians. Many of these people don’t know what tools are available to scale up and scale out,” Davis continues. “Building awareness in the life sciences community for new technology is critical to the success of these tools. We need to advertise how new automation technology can advance cell therapy, gene therapy and cell biology in general.”
To introduce new technology in this life sciences area, Puleo, Smith and Davis served as guest editors for the August 2018 SLAS Technology Special Issue on Enabling Technology in Cell-Based Therapies: Scale-Up, Scale-Out or Program In-Place. The trio secured articles featuring technologies that promise to change the way in which researchers and clinicians process and use therapeutic cells.

“These are tools we could see ourselves using,” says Puleo, a member of the SLAS Technology Editorial Board and one of the researchers included in the 2018 SLAS Technology Ten, which honors innovative scientific achievements published in SLAS Technology the previous year. “I love to build tools, diagnostics or therapies that go into devices to directly help patients. That’s what excites me about participating in this special issue – the potential to improve treatments for patients. To do this, we need to improve cell therapy manufacturing,” he continues. “The new, innovative technologies in cell-based therapies featured in the special issue show promise for scale and integration into the whole work flow.”
Puleo’s hope is that readers of the special issue will consider the importance of bringing new technology into their labs to decrease the cost and increase the integration in cell manufacturing workloads. “In five years, if these technologies are interfaced together and work within one work flow to decrease the costs of future cell therapies and the risks involved, then we’ve done our jobs,” he says.
Davis thinks that as the technologies get adopted, “the cost of manufacturing will come down. To put it in perspective, the cost of many cell therapies to the patient is hundreds of thousands of dollars if not higher because the upstream processes are so expensive,” he says.
Smith feels that some of the onus to move forward lies with the content developers. “The sooner the content developers accelerate their efforts and thereafter the commercial adaption rate of their developments, the better and more robust manufacturing process will become,” he says, adding that scaling up and out to robustly manufacture cell therapies is challenging.
“Cell and gene therapies rely on the core science of cell biology,” Smith explains. “Many researchers use long-established tools in the cell therapy research space that were designed for research. They are not necessarily scalable for commercial manufacture.
“The approach taken to get to this point, especially relating to autologous stem cells, or therapy where cells are taken from a patient and returned to the same patient, is to use ad hoc instrumentation that is readily available, such as blood banking centrifuges and bioreactors,” continues Smith, who has developed cell therapy tools and reagents for GE for more than seven years. “Those borrowed tools were good to get to proof of principal, make enough doses to get through trials, and even to get started at low volume quantities in commercial manufacturing, post approval. However, as more indications are approved, the throughput scale manufacturing demand will be limited by that kind of infrastructure.”
Smith would like to see practitioners in academia and government labs move beyond the tried-and-true, decades-old open systems and explore new technology. “These innovators are propelling this space forward from clinical trials to approval and commercial success,” he says. “Now is the time to put new types of purpose-built tools into this space."
Seven articles compose the SLAS Technology special issue, with two reviews and five original research articles that span a diversity of technologies, cell types and methods that Davis appreciates. “The choice of cell types represented in these papers is a nice cross section of the different work being done from pluripotent stem cells to T-cell therapies to organoids,” he says.
The two review articles included in the SLAS Technology special issue detail the status of cell bioreactors in both stem cell and tissue/organ engineering applications. "Automated Cell Culture Systems and Their Applications to Human Pluripotent Stem Cell Studies," from researchers at the Centre for Eye Research Australia (East Melbourne, Australia), provides an overview of systems currently available for automated stem cell culture and reviews the capacities, capabilities and relative limitations of each. "Tissue and Organ 3D Bioprinting," from researchers at the Department of Biomedical Engineering, Center of Biomanufacturing for Regenerative Medicine, Watson School of Engineering and Applied Science, Binghamton University, State University of New York (Binghamton, NY, USA), discusses the state of 3D bioprinting, along with a broad range of biomaterials used for it.
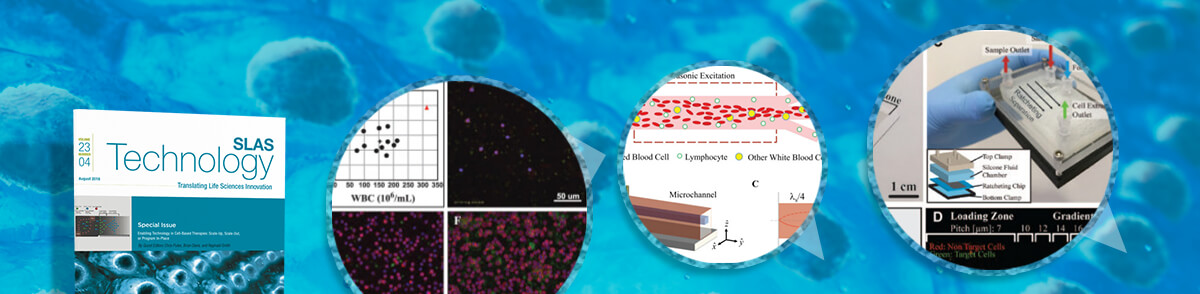
Puleo is particularly enthusiastic about the original research articles that focus on separation techniques for selecting and monitoring cells and the potential for progress shown in this research. “Research groups that contributed to the special issue are showing scale results that are relevant to cell manufacturing, taking fluid from microliters to liters at very rapid pace for the first time,” says Puleo. “We’ve seen these cell separation techniques – using magnetic, acoustic and microfluidic tools – in the past, but in a very small scale with microliter volumes processed at a very low-thoughput.”
The article highlighting a magnetic tool, “Continuous and Quantitative Purification of T-Cell Subsets for Cell Therapy Manufacturing Using Magnetic Ratcheting Cytometry,” reports on a continuous-flow, quantitative cell enrichment platform based on a technique known as ratcheting cytometry that can perform complex, multicomponent purification targeting various subpopulations of magnetically labeled T cells directly from apheresis or peripheral blood mononuclear cell (PBMC) samples. The authors, from the Department of Bioengineering at the University of California, Los Angeles, believe that ratcheting cytometry’s quantitative capacity and throughput scalability represents an excellent technology candidate to alleviate cell therapy manufacturing bottlenecks.
An acoustic tool featured in “Purification of Lymphocytes by Acoustic Separation in Plastic Microchannels” presents an alternative approach to blood cell purification using acoustic forces in plastic microchannels. The research team at The Charles Stark Draper Laboratory (Cambridge, MA, USA) expects that future scale-up of their system for increased throughput will enable its incorporation into cell therapy workflows, and that it could ultimately reduce costs and expand access for patients.
“Deterministic Lateral Displacement: The Next-Generation CAR T-Cell Processing?” represents a microfluidic separator, and in this research, scientists from GPB Scientific (Richmond, VA, USA) extend their previous work in whole blood by manufacturing a highly parallel deterministic lateral displacement (DLD) device incorporating diamond microposts and moving into processing, for the first time, apheresis blood products. DLD processing provides a path to develop a simple closed system that can be automated while simultaneously addressing multiple steps when there is potential for human error, microbial contamination and other current technical challenges associated with the manufacture of therapeutic cells.
A unique aspect that these articles present, says Puleo, is technology that performs both cell separation tasks and quality control and analysis tasks. “Some of the methods used for separation are also informing the size, type and activity of the cell and I think that will be important in the near future,” he comments.
The special issue also includes two articles in which the guest editors, along with their research teams at GE, describe methods to better automate or package complex cell manipulations into closed bioreactor systems. “High-Capacity Redox Polymer Electrodes: Applications in Molecular and Cellular Processing” presents methods to fabricate high-capacity redox electrodes, using thick membrane or fiber casting of conjugated polymer solutions, that may be built directly into processing systems for applications such as electrical cell lysis and electrokinetic molecular separations. “Automated Closed-System Expansion of Pluripotent Stem Cell Aggregates in a Rocking-Motion Bioreactor” examines a low-shear method for the expansion of cells in a rocking-motion bioreactor. The rocking motion drives continual mixing and aeration, and the single-use disposable bioreactors avoid issues around contamination during seeding, medium exchange, passage and cell harvest.
Smith describes some of the tools – i.e., DLD and acoustic microfluidic separation platforms, as well as magnetic beads coupled to ratcheting cytometry – included in the special issue as component technologies for cell isolation. “There is great potential for these tools to be integrated into larger more complete cell processing platforms that need to emerge as the cell therapy space moves through regulatory approval into commercial production and robust manufacturing,” he comments. “The way these components work helps crystalize for us how these platforms develop, the functionalities they possess and the kind of production scales they support. I think it’s important to understand the limits and the capabilities of these technologies.”
The SLAS Technology Special Issue is available now at SLAS Technology Online for SLAS members, SLAS Technology subscribers and pay-per-view readers. Free public access becomes available one year after final (print) publication.
UCLA’s Coleman Murray, Ph.D., talks about his contribution to the August 2018 special issue of SLAS Technology on Enabling Technology in Cell-Based Therapies, by guest editor Chris Puleo and his associates at General Electric Global Research, in a 2018 SLAS Technology podcast. Murray’s article illustrates an important example of how new technologies are changing automation and processing in cell therapy manufacturing.
Cancer Research Institute Reports on the Global Landscape of Cancer Cell Therapy
Read More about the People Powering the 2018 SLAS Technology Ten in the SLAS E-Zine
Learn More about Chris Puleo's Mesodissection Platform in SLAS Technology